 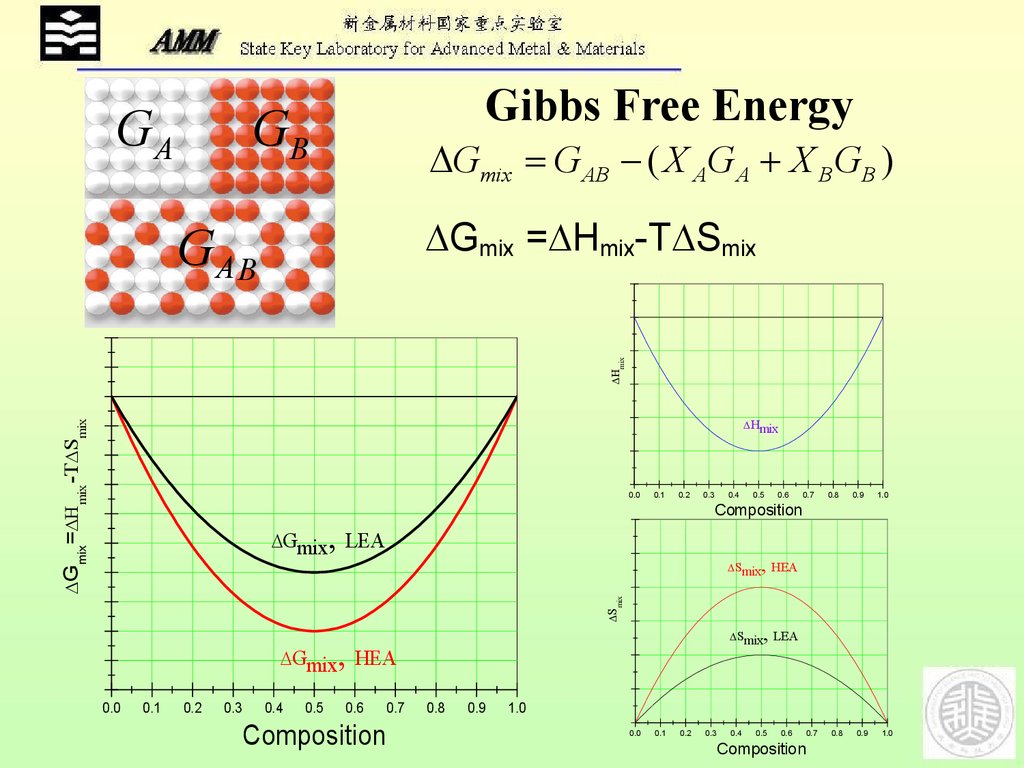
Equation (6) is utilized to describe the mixture of ideal gases 15-the textbook scenario for an ideal mixture-and by Gaskell and Laughlin 16 to introduce ideal solutions in the solid state. In an ideal solution, the mixing enthalpy is per definition zero. In chemical thermodynamics, such mixing scenarios are referred to as ideal solutions. (3), only purely random mixing scenarios can be described. 9 At this point, it is evident that with w, as defined in Eq. “This concept of trying to improve magnetic properties through Spinodal Decomposition is very new,” says Dutta, “And these new mechanisms will help us to find new magnetic material for potential use in, for example, refrigeration systems based less on gases and more on solid-state magnetic materials which will be much more environmentally friendly.This equation was utilized by Yeh 9 to compute the change in molar configurational entropy of HEAs and to define HEAs in terms of a critical-or threshold-entropy value. With a better understanding of these mechanisms, researchers can begin to investigate other magnetic HEAs and multicomponent alloys to determine whether this same behaviour occurs causing an improvement in their magnetic properties. So in fact there are two mechanisms at work here: one is the formation of two regions with different chemical compositions - a phenomenon known technically as Spinodal Decomposition - and the other factor is the resulting difference in volume and therefore coherency stress between the different regions. “And if one of these regions is particularly important for the magnetic properties, a volume expansion can improve those magnetic properties.” Using this information, Dutta ran theoretical simulations and was ultimately able to explain why, after prolonged heating, you get improved magnetic properties: “Copper doesn’t like to make a solid homogenous mixture with the other elements and so the more you heat the sample, the more the copper tries to separate out from the other four elements, leading to different regions with different compositions - for example, an iron-cobalt rich region and a copper rich region.” These different regions have unequal volumes causing what is known as coherency stress between a bigger volume and a smaller one. And two, that within the material, the different elements became segregated into different regions within the alloy.” “And they noticed two things: one was that heating the HEA for 240 hours improved its magnetic properties.  
“Our colleagues at the Max-Planck-Institut für Eisenforschung in Germany heated this material at a particular fixed temperature for different lengths of time,” says Dutta. In this study, the team played around with the composition of a FeCoNiMnCu HEA, which contains iron, cobalt, nickel, manganese and copper. steel, which is an alloy of iron mixed with 1% carbon, HEAs consist of five or more elements in more-or-less equal proportions. Unlike traditional alloys, which usually consist of one major component with a small amount of another added element e.g. “And to do this, you have to play with the chemistry so change the composition of the alloy.” “The focus of this project was to find new mechanisms with which we could improve the magnetic properties of an HEA,” says Dutta. greater strength, promising magnetic properties, and better resistance to rusting and corrosion. High-Entropy Alloys (HEAs) were first proposed about 15 years ago and since then, they have aroused a lot of interest within the materials science community because of their excellent physical, mechanical and functional properties e.g. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
